Hunger hormones control the degree to which individuals sense hunger and fullness or, satiety, and can contribute to weight loss and maintenance success. Research shows that a significant percentage of dieters regain most, if not all, of the weight they shed within a year, a factoid every personal trainer should be cognizant of.
Increased appetite/loss of willpower top of the list of common reasons. This is why it is vital to consider the impact of hunger hormones on eating habits. To help clients who face this dilemma after months of successful weight loss, read on and learn about hormones responsible for hunger and satiety, and how they can easily turn against a serious dieter.
Duality of Satiety Hormones
The control of energy balance within the human body largely rests on two key hormones, leptin and ghrelin. Leptin, produced by adipose tissue, mediates satiety/appetite and long-term regulation of energy balance. This hunger hormone works to suppress food intake, letting the body know enough fuel has been ingested, and to begin the fat-burning creation of energy. This in turn kickstarts the weight loss process.
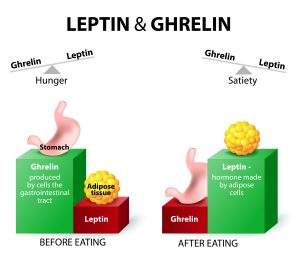
Ghrelin and leptin – hormones regulating appetite. Leptin the satiety hormone. Ghrelin the hunger hormone. When ghrelin levels are high, we feel hungry. After we eat, ghrelin levels fall and we feel satisfied.
Ghrelin, a faster-acting hunger hormone released by an empty stomach, plays a role in meal initiation/short-term food intake. It measures the highest right before consumption, and dips maximally within 60 minutes following a meal.
“In a perfectly working body, ghrelin tells us to eat so we don’t die of starvation, and leptin tells us when to stop,” says Dr. Michelle Sands, hormone, metabolism, and epigenetics expert, and author of Hormone Harmony over 35. Unfortunately, hormones sometimes find that perfect balance elusive.
“Leptin is a bigger player than ghrelin when it comes to weight gain and energy balance,” Dr. Sands says. “It’s closely tied to your thyroid and brain. When leptin is working well, we have a better metabolic rate, mood regulation, memory, brain function, and mental sharpness. When it’s not, it can play a role in obesity, mood swings, and brain fog.”
In addition, Dr. Sands says that, “Some people are genetically predisposed to release more ghrelin, and they get hungry faster as a result.”
Anorexia and Hunger Hormone Dysregulation
Research highlights how eating disorder sufferers have problems regulating feelings of hunger and satiety; whether this hails from an emotional or physiological place, such circumstances may perpetuate and/or contribute to the onset of anorexia.
Given that leptin directly affects appetite/body weight regulation, scientists set out to determine whether leptin played any role in eating disorders. Researchers measured leptin levels in 67 women: 21 anorexics, 32 bulimics, and 14 binge eaters. They compared/contrasted these values to those of 25 healthy women. Not surprisingly, the anorexic subjects displayed much lower leptin levels.
As anorexia proceeds to a more serious level, ghrelin levels go up, as expected with someone hovering near starvation. Upon the refeeding that accompanies intense therapy, the level of ghrelin plummets, which may explain why many people with anorexia struggle to put on weight. After long-term bouts of the disease, normal hunger signals frequently fail to function. It requires time and a tremendous caloric load to add weight safely.
Why Extremely Low-Calorie Diets Fail
The physiological status of an anorexic patient differs vastly from that of an average dieter; therefore, where these individuals seem to have found a way – albeit dangerous – to drop significant weight and keep it off, those results do not necessarily apply across the board.
Researchers at Rockefeller University discovered the leptin gene almost 20 years ago. We have now come to understand how leptin functions as one of many reasons why diets don’t work. As body fat stores go down, so too do circulating leptin levels. Lower leptin levels throw off the body’s satiety response; now the body takes longer to register the feeling of fullness following a meal. Over time, this scenario serves to bring the body back to its original weight, often with a higher percentage of fat tissue than before the loss.
Leptin levels aren’t the only contributing factor. A group of researchers in the United Kingdom hypothesized that an enzyme called glucokinase might play some part in our desire for consuming glucose. The enzyme serves to sense glucose in the liver and pancreas, but it is also present in the hypothalamus, a part of the brain that regulates a variety of vital functions including food intake.
It’s long been theorized that glucose-rich foods would not have been as easily found in the past, leading to an innate mechanism to seek them out in preference to others. With that in mind, the researchers devised an experiment using rats, which, like humans, have been found to consume glucose in preference to other foods. The scientists found that when the animals haven’t eaten for 24 hours, the activity of glucokinase in a part of the hypothalamus that controls appetite increased significantly.
In the experiment, the rats were given access to both a glucose solution and their regular “chow” diet of food pellets. The researchers used a virus in one group to increase the activity of glucokinase in the hypothalamus, causing those rats to consume more glucose than chow. The scientist noted that when glucokinase activity was decreased, the rats consumed less glucose.
The results, the researchers say, suggest that there may be a mechanism in the hypothalamus that can detect how much glucose is reaching the brain, triggering a hunger for carbohydrates when animals detect there isn’t enough present. The study is believed to be the first of its kind to identify a mechanism in the brain that responds to a specific nutrient, rather than to a shortfall in overall energy intake.
One implication is that the types of nutrients we consume could be at least as important as the number of calories we consume. This, in turn, might mean it is possible to cut down on cravings for glucose-rich foods and beverages by changes in diet. Because glucokinase levels vary from person to person, however, there may be no one-size-fits-all formula.
Pleasure Reward of Food
The function of ghrelin first gained notice as a stomach-derived hunger hormone involved in energy balance, hunger, and food consumption. Recent work now highlights its role in reward-driven behavior via activation of the so-called “cholinergic-dopaminergic reward link”.
According to research published in the journal Addiction Biology, the hunger hormone ghrelin not only reduces fat utilization but also plays a key role in the food reward cascade controlled by the brain’s pleasure-reward system. Ghrelin levels are negatively correlated with weight, so dieting (especially severe calorie restriction) tends to increase ghrelin output.
Paradoxical Effects of Dieting
Individuals who embark upon severe caloric restriction to reduce weight often find the diet cannot be sustained long-term, due to an eventual lack of willpower, increased hunger, and a reduction in their metabolic rate. The human body interprets such a situation as “famine” and reacts appropriately for survival. As metabolism slows to conserve energy, dieters very often experience a surprising regaining of weight.
Leptin levels fall rapidly in response to fasting/severe caloric restriction, thereby bringing about profound hormonal changes. Low leptin levels can lead to overfeeding and suppressed thyroid activity.
Stress-Eating, Hunger Hormones, and Fat Levels
In addition to increasing appetite, ghrelin may also encourage the build-up of abdominal fat. Increased ghrelin syncs directly with stressful situations, as evidenced by the multitude of individuals who tend to mindlessly eat when stressed. Ghrelin contributes to weight gain by maintaining these stress levels (rather than allowing the body to return to homeostasis); the cascade culminates in strong urges to randomly snack or overeat.
On The Horizon
Several molecules and signaling pathways associated with leptin and ghrelin receptors have been identified as potential targets for pharmaceutical treatment, with the goal of overriding the biological resistance to these hormones. Developing such medications could someday mean more effective treatments for individuals living with obesity.
Until then, we can help foster healthier lifestyles for our clients by steering them away from fad diets, and more towards prudent eating habits. Balancing macronutrients and exercise, trainers and dietitians can help clients conquer the mysteries of hunger hormones like leptin and ghrelin and thereby gain better control over their physiques.
References
www.healthline.com/nutrition/ghrelin
www.ncbi.nlm.nih.gov/pmc/articles/PMC3901982/
www.ncbi.nlm.nih.gov/pmc/articles/PMC4443295/
www.winchesterhospital.org/health-library/article?id=37216
www.ncbi.nlm.nih.gov/pmc/articles/PMC2430504/
pubmed.ncbi.nlm.nih.gov/16759336/
pubmed.ncbi.nlm.nih.gov/21354264/
www.thehealthsite.com/fitness/diet/how-you-can-reduce-your-hunger-hormone-ghrelin-k1217-544991/
www.nationaleatingdisorders.org/toolkit/parent-toolkit/appetite
www.nature.com/articles/nrendo.2016.222
www.endocrineweb.com/news/63844-how-hunger-hormones-control-weight-loss
Syed Hussain, Errol Richardson, Yue Ma, Christopher Holton, Ivan De Backer, Niki Buckley, Waljit Dhillo, Gavin Bewick, Shuai Zhang, David Carling, Steve Bloom, James Gardiner. Glucokinase activity in the arcuate nucleus regulates glucose intake. Journal of Clinical Investigation, 2014; DOI: 10.1172/JCI77172
pubmed.ncbi.nlm.nih.gov/14677575/
pubmed.ncbi.nlm.nih.gov/15265818/
pubmed.ncbi.nlm.nih.gov/17212793/
Cathleen Kronemer is an NFPT CEC writer and a member of the NFPT Certification Council Board. Cathleen is an AFAA-Certified Group Exercise Instructor, NSCA-Certified Personal Trainer, ACE-Certified Health Coach, former competitive bodybuilder and freelance writer. She is employed at the Jewish Community Center in St. Louis, MO. Cathleen has been involved in the fitness industry for over three decades. Feel free to contact her at trainhard@kronemer.com. She welcomes your feedback and your comments!


